Artificial Intelligence and Drug Development
IMPORTANT DISCLOSURE: This article is provided for general informational and educational purposes only. It does not constitute investment advice, a solicitation, or a recommendation to buy or sell any security or investment product. The securities and companies referenced are for illustrative purposes only and do not represent a complete analysis or a recommendation. Past performance is not indicative of future results. All investments involve risk, including the possible loss of principal. Readers are strongly encouraged to consult a qualified financial advisor or registered investment adviser before making any investment decisions. Nothing in this article creates a fiduciary or advisory relationship of any kind.
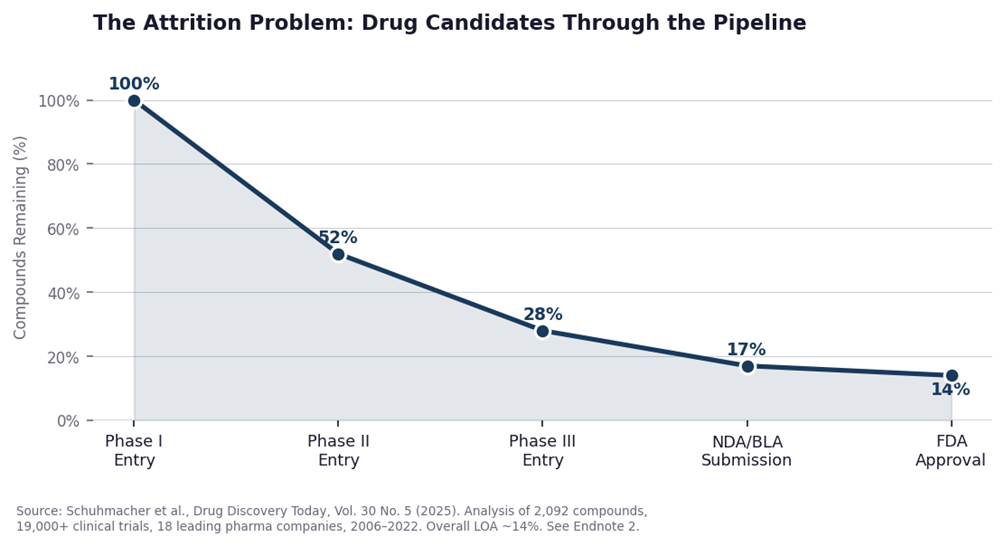
Drug development is, by almost any measure, one of the most expensive and failure-prone endeavors in modern industry. When you factor in all the failed attempts along the way, it costs roughly $2.6 billion and takes more than a decade.1 And even after all that, most drugs never make it. Research shows that only about 10% to 14% of drug candidates that enter clinical testing ultimately win approval.2
As a potential way to change those odds, artificial intelligence has attracted substantial institutional capital, major pharmaceutical partnerships, and a growing body of peer-reviewed research. AI tools are now being used at multiple stages of the drug development process, from the initial search for promising molecules all the way through clinical trial design. At the same time, the central question, whether AI-assisted drugs perform better in human clinical trials, remains unanswered. That uncertainty is material to any investment assessment in this space.
The Problem AI Is Trying to Solve
To understand why AI matters here, it helps to know why drug development is so expensive in the first place. The Tufts Center for the Study of Drug Development, a research institution that works with the pharmaceutical industry, estimates that bringing a single new drug to market costs around $2.6 billion once you include the cost of all the projects that fail along the way.1 Some researchers argue that figure is inflated by the way the industry accounts for its costs3, but the core point is not seriously disputed: drug development is expensive and most attempts fail.
Just how often do they fail? A 2025 study in the journal Drug Discovery Today looked at more than 2,000 drug compounds and nearly 20,000 clinical trials run by 18 major pharmaceutical companies from 2006 to 2022. It found that only about 14% of drugs that entered clinical trials ever won FDA approval.2
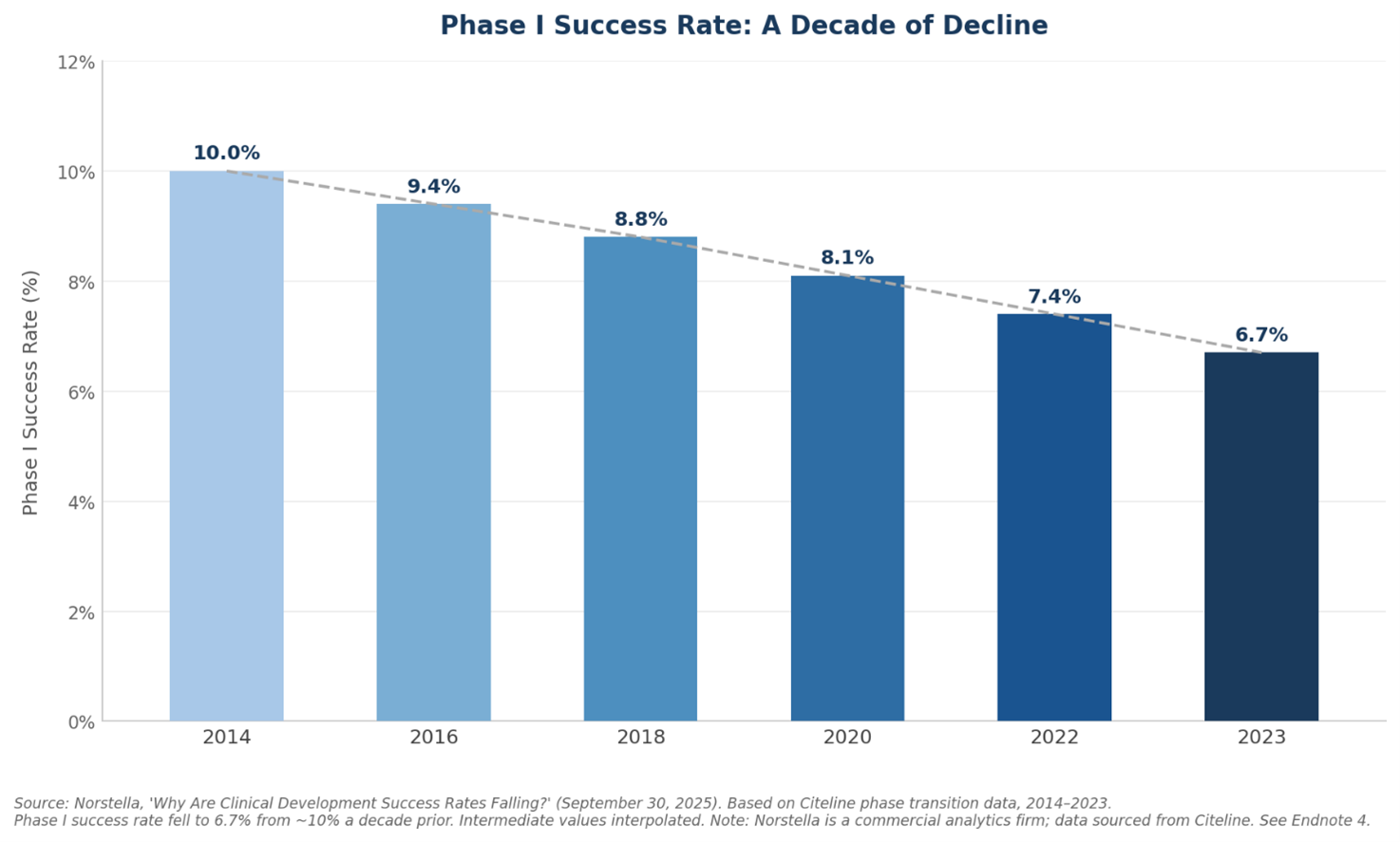
The trend is also moving in the wrong direction. Phase I success rates have fallen by a third over the past decade. A separate industry analysis found that over the ten-year period through 2023, the average likelihood of a drug entering Phase I ever reaching approval had fallen to 6.7%, down from around 10% a decade earlier.4 That failure rate shapes everything about how drug companies operate. They have to fund many candidates at once, knowing most will not pan out, because the ones that do succeed need to cover the cost of all the ones that did not. AI is being applied primarily in the early stages of this process, where better tools for identifying promising candidates could meaningfully reduce waste and improve the odds downstream.
What AI Is Actually Doing in Drug Discovery
AI in drug development is not one single tool. It is a collection of different technologies being applied to different parts of the process. Three applications have drawn the most interest.
Predicting protein shapes. Proteins are the tiny biological machines that carry out most of the work in your body. Many diseases are caused by proteins that are not working correctly. To design a drug that fixes a broken protein, scientists first need to know exactly what shape that protein takes in three dimensions. Until recently, figuring that out could take a research team months or even years for a single protein. In 2020, an AI system called AlphaFold, built by Google DeepMind, demonstrated protein shape prediction accuracy that, according to Google DeepMind, rivaled experimental methods in a matter of hours. By 2022, Google DeepMind and the European Bioinformatics Institute had made a database of over 200 million predicted protein structures freely available to scientists worldwide.5 AlphaFold's creators were awarded the Nobel Prize in Chemistry in 2024.
Designing new molecules. Traditional drug discovery involves searching through large libraries of existing chemical compounds to find ones that might work as a treatment. AI generative models can do something more ambitious: propose entirely new molecular structures that have never existed before, designed from the ground up to hit a specific target. Some companies have built meaningful parts of their businesses around this capability.
Improving clinical trials. AI is also being used to help design better clinical trials. That includes identifying which patients are most likely to benefit from a given drug, predicting potential side effects before human testing begins, and flagging design improvements that raise the chances of a clear result. Peer-reviewed research has examined AI applications across all stages of trial risk assessment, including patient safety, efficacy prediction, and operational efficiency.6
One important distinction is worth keeping in mind. AI has shown clear value in the early, lab-based stages of drug discovery.5 Whether that translates into better outcomes in human clinical trials is a separate and harder question, and the answer is still being written.
Where Investment Activity Is Concentrated
Most major pharmaceutical companies have announced AI-related deals or partnerships, and significant venture capital investment has flowed into this space in recent years.
Large pharma companies using AI. These are established businesses with steady revenues, many paying dividends, and decades of drug development experience. AI is being added to what they already do, not replacing it. A recent example: in January 2026, Eli Lilly and Nvidia announced a joint AI co-innovation lab in the San Francisco Bay Area, backed by a commitment of up to $1 billion over five years.7 The lab will bring together Lilly's biology and scientific teams alongside Nvidia's AI engineers to work on accelerated drug discovery, clinical trial optimization, and advanced manufacturing. This deal reflects the scale at which some large companies are integrating AI into their drug development operations.
Pure-play AI drug discovery companies. These are companies whose entire focus is using AI to find and develop drugs. Many are not yet profitable, and their stock prices depend heavily on whether their pipeline candidates eventually succeed in clinical trials. The historical failure rate in drug development is relevant context for any assessment of these companies. Companies in this category include Recursion Pharmaceuticals (RXRX) and Schrodinger (SDGR), among others.8
Technology infrastructure companies. Semiconductor companies like Nvidia and cloud providers like Microsoft and Amazon supply the computing power that makes AI work across every industry, including drug development. Nvidia’s partnership with Eli Lilly is a concrete example: the two companies are also building what Lilly describes as the most powerful supercomputer owned by any pharmaceutical company, powered by more than 1,000 Nvidia GPUs and designed to serve as an “AI factory” running the full cycle from data to drug candidate.7 Because these infrastructure companies serve many markets well beyond pharma, they are not as directly tied to any single drug succeeding or failing. That said, their valuations reflect many factors beyond drug discovery.
Risks Worth Taking Seriously
The gap between the lab and the clinic. AI has delivered real progress in the early parts of drug discovery, especially in protein prediction and molecule design. But predicting whether a drug will be safe and effective in actual human beings is a much harder problem, and AI has not solved it yet. Most AI-assisted drug candidates have not yet produced Phase III clinical data, which is the final hurdle before approval. High failure rates in drug development are not mainly an information problem. They often come down to biological complexity that no model has fully figured out.
Valuation risk. Many pure-play AI drug companies were valued very richly during the 2020 to 2022 technology boom, based on hopes about future drugs rather than current sales. Markets have pulled back from those peaks, but some companies still trade at substantial premiums relative to where they actually are today. If clinical results disappoint, or if the broader investment climate tightens, these stocks can fall hard and fast.
Regulatory uncertainty. The FDA is still working out exactly how it will evaluate drugs that were developed primarily using AI. Until that guidance is in place and battle-tested, there is some real uncertainty about approval timelines and what data requirements might look like for AI-assisted candidates.
Competition and geopolitics. This field is heavily funded globally, with strong programs in the U.S., Europe, and China. A technology advantage today does not guarantee an advantage tomorrow. The international dimension also brings uncertainty around intellectual property and data-sharing rules.
What the Next Few Years Could Look Like
A number of AI-assisted drugs are currently in late-stage clinical testing. Insilico Medicine completed a Phase IIA trial for a drug targeting idiopathic pulmonary fibrosis developed using generative AI. The results were published in Nature Medicine in June 2025 and represented one of the first peer-reviewed clinical validations of an AI-generated drug candidate.9 Recursion Pharmaceuticals and several other companies have multiple candidates moving through the clinical pipeline.10
Over the next several years, we will start to get meaningful Phase III data on AI-assisted drugs, and that will be the real test. If AI-assisted drugs begin producing positive Phase III results, that would likely draw increased investment attention to the sector, as has generally been the pattern following clinical breakthroughs in other areas. A run of failures would trigger a reassessment of timelines and valuations. Either way, the clinical data coming out of these trials will be more instructive than current market sentiment.
There are also adjacent areas worth watching. Personalized medicine, which tailors treatment to individual patients based on their genetic profiles, is an area where AI’s ability to work through large, complex datasets is increasingly being applied. Drug repurposing, which is identifying new uses for already-approved drugs, is another area where AI is active, and one that may reduce some of the time and cost involved compared to developing a new compound from scratch.11
Conclusion
AI is being applied to drug discovery in ways that address real and long-standing problems in how new medicines get developed. The work on protein structure prediction is backed by strong science and has been recognized at the highest levels, including the Nobel Prize. The harder question, whether AI-discovered drugs will work at scale in human patients, is one the industry is still in the process of answering. Clinical failure rates remain high, the regulatory framework is still taking shape, competition is intense, and in some parts of this sector, valuations continue to price in outcomes that the clinical data has not yet supported. These are not minor footnotes to an otherwise compelling story but are central to any honest assessment of this space.
ENDNOTES
1. DiMasi, Joseph A., Henry G. Grabowski, and Ronald W. Hansen. "Innovation in the Pharmaceutical Industry: New Estimates of R&D Costs." Journal of Health Economics 47 (2016): 20-33. Note: This figure is derived from industry-provided data and is contested by some researchers; see Endnote 3 for an alternative perspective.
2. Schuhmacher, Alexander, Markus Hinder, Elazar Brief, Oliver Gassmann, and Dominik Hartl. "Benchmarking R&D Success Rates of Leading Pharmaceutical Companies: An Empirical Analysis of FDA Approvals (2006-2022)." Drug Discovery Today 30, no. 5 (2025): Article 104326.
3. “R&D Cost Estimates: MSF Response to Tufts CSDD Study on Cost to Develop a New Drug." Medecins Sans Frontieres Access Campaign, November 2014. Available at: msfaccess.org.
4. Norstella. "Why Are Clinical Development Success Rates Falling?" September 30, 2025. norstella.com. Based on Citeline phase transition data covering the 10-year period from 2014 to 2023. Reports average likelihood of approval from Phase I at 6.7%, described as an all-time low. Phase II remains the single largest attrition point, with only 28% of programs advancing. Note: Norstella is a commercial analytics firm; underlying data sourced from Citeline, an established pharmaceutical intelligence database used widely across the industry.
5. AlphaFold Protein Structure Database. EMBL-EBI and Google DeepMind. alphafold.ebi.ac.uk. Database expanded in July 2022 to over 200 million predicted protein structures. See also: Jumper, John, et al. "Highly Accurate Protein Structure Prediction with AlphaFold." Nature 596 (2021): 583-589. Nobel Prize in Chemistry 2024 awarded to Demis Hassabis and John Jumper (Google DeepMind) and David Baker (University of Washington).
6. Teodoro, Douglas, et al. "A scoping review of artificial intelligence applications in clinical trial risk assessment." npj Digital Medicine (Nature), July 30, 2025. doi: 10.1038/s41746-025-01886-7. Review of 142 peer-reviewed studies published 2013–2024 covering AI applications in safety, efficacy, and operational risk prediction in clinical trials.
7. NVIDIA and Eli Lilly and Company. "NVIDIA and Lilly Announce Co-Innovation AI Lab to Reinvent Drug Discovery in the Age of AI." Press release, J.P. Morgan Healthcare Conference, January 12, 2026. investor.nvidia.com. The co-innovation lab will be located in the San Francisco Bay Area. The related supercomputer partnership was announced October 28, 2025. investor.lilly.com. Eli Lilly and Nvidia are referenced for illustrative purposes only and do not represent a recommendation.
8. Recursion Pharmaceuticals (RXRX) and Schrodinger (SDGR) are referenced as illustrative examples only. These references do not constitute a recommendation to purchase, hold, or sell these or any other securities. Investors should conduct independent due diligence and consult a qualified financial advisor before making investment decisions.
9. Ren, Feng, et al. "A generative AI-discovered TNIK inhibitor for idiopathic pulmonary fibrosis: a randomized phase 2a trial." Nature Medicine, June 3, 2025. doi: 10.1038/s41591-025-03743-2. This trial is among the first peer-reviewed Phase IIa results published for an AI-generated drug candidate. Insilico Medicine was the study sponsor.
10. Recursion Pharmaceuticals, Inc. Pipeline and corporate update. SEC filing, 2024 Annual Review. Available at: sec.gov. Pipeline includes multiple Phase 1/2 and Phase 2 programs across oncology and rare disease indications. Referenced for illustrative purposes only and does not constitute a recommendation.
11. Wan, Z., et al. "Applications of Artificial Intelligence in Drug Repurposing." Advanced Science (Wiley), 2025. doi: 10.1002/advs.202411325. The authors note that repurposing already-approved compounds can bypass early development stages, potentially reducing associated time and cost.
DISCLOSURES AND IMPORTANT INFORMATION
This article is provided by Chatham Capital Group for general informational and educational purposes only. It does not constitute investment advice, financial planning advice, legal advice, tax advice, or a solicitation or offer to buy or sell any security or investment product. This material has not been tailored to the investment objectives, financial situation, risk tolerance, or specific needs of any individual reader.
The information in this article reflects the views and research of the author as of the date of publication and is subject to change without notice. No representation is made that the information is accurate, complete, or current. Past performance of any investment strategy, security, or sector is not indicative of future results. All investments involve risk, including the possible loss of the entire principal amount invested.
References to specific companies and securities, including Recursion Pharmaceuticals (RXRX), Schrodinger (SDGR), Nvidia, Microsoft, Amazon, Pfizer, Merck, AstraZeneca, Novartis, and Insilico Medicine, are for illustrative and educational purposes only. They do not represent a complete or current analysis of any security and should not be interpreted as a recommendation or endorsement to purchase, hold, or sell any security. Investors should conduct their own due diligence before making any investment decision.
Nothing in this article creates a fiduciary, advisory, or client relationship of any kind. Readers are strongly encouraged to consult with a qualified financial advisor or registered investment adviser before making any investment decisions. Chatham Capital Group's Form ADV Part 2A (Brochure) is available through the SEC's Investment Adviser Public Disclosure database at www.adviserinfo.sec.gov.
This material is not an advertisement for investment advisory services. Chatham Capital Group is a registered investment advisory firm headquartered in Savannah, Georgia.